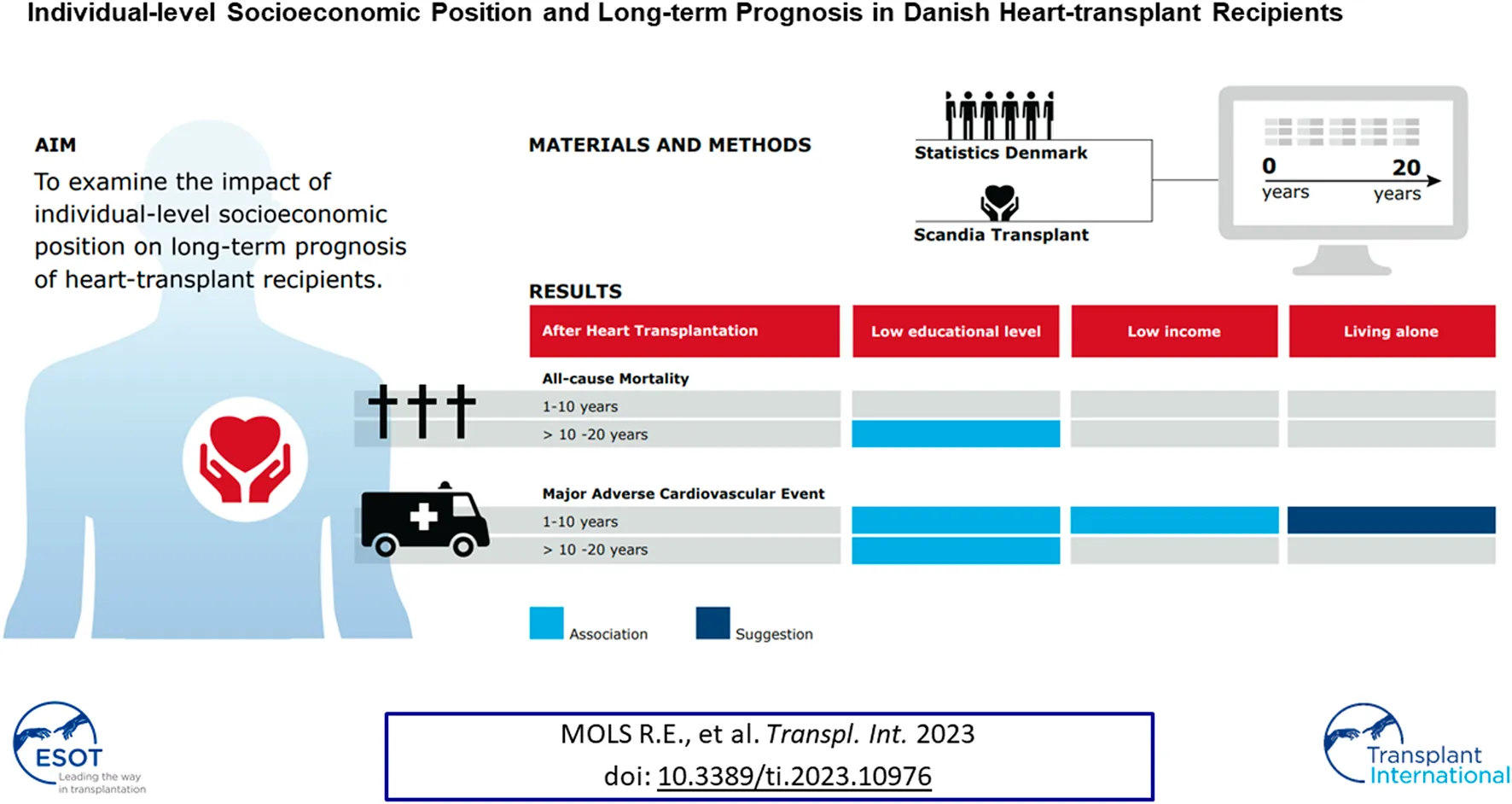
Abstract
Socioeconomic deprivation can limit access to healthcare. Important gaps persist in the understanding of how individual indicators of socioeconomic disadvantage may affect clinical outcomes after heart transplantation. We sought to examine the impact of individual-level socioeconomic position (SEP) on prognosis of heart-transplant recipients. A population-based study including all Danish first-time heart-transplant recipients (n = 649) was conducted. Data were linked across complete national health registers. Associations were evaluated between SEP and all-cause mortality and first-time major adverse cardiovascular event (MACE) during follow-up periods. The half-time survival was 15.6 years (20-year period). In total, 330 (51%) of recipients experienced a first-time cardiovascular event and the most frequent was graft failure (42%). Both acute myocardial infarction and cardiac arrest occurred in ≤5 of recipients. Low educational level was associated with increased all-cause mortality 10–20 years post-transplant (adjusted hazard ratio [HR] 1.95, 95% confidence interval [CI] 1.19–3.19). During 1–10 years post-transplant, low educational level (adjusted HR 1.66, 95% CI 1.14–2.43) and low income (adjusted HR 1.81, 95% CI 1.02–3.22) were associated with a first-time MACE. In a country with free access to multidisciplinary team management, low levels of education and income were associated with a poorer prognosis after heart transplantation.
Introduction
Heart transplantation is a widely accepted procedure improving survival, quality of life, and physical capacity in patients with end-stage heart failure (, ). During the past 30 years, survival rates have increased significantly, despite high-risk and older recipients undergoing heart transplantation (, ). Currently, the 50% survival estimate after heart transplantation in adults is 12.5 years, and 14.8 years when conditional on 1-year survival (). Advances in immunosuppressive treatment and perioperative care have improved 1-year survival to approximately 90% (). The main causes of death immediately following heart transplantation are primary graft dysfunction, rejection, and infection; primary causes of long-term mortality are cardiac allograft vasculopathy, non-specific graft failure, renal dysfunction, and malignancy (, ). It is pivotal for follow-up of heart transplant recipients that transplant centers establish multidisciplinary team management programs, designed to improve survival (, ).
Studies in both the United States and the United Kingdom have shown that multiple indicators of index-based socioeconomic position (SEP) are associated with death, independent of baseline clinical characteristics of heart transplant recipients (–). Among American heart transplant survivors, low SEP (score) predicted an increased risk of rejection and graft loss (). Earlier studies in the United States have suggested higher mortality in patients covered by Medicare compared with patients covered by private insurance at the time of heart transplantation (, ). Studies primarily conducted in the United States have also reported that depression before or early after heart transplantation is associated with higher post-transplant mortality (–). Mental health conditions often coexist with physical chronic diseases (). Multimorbidity including chronic psychiatric disorder has been associated with higher mortality (, ). Moreover, data support a strong socioeconomic gradient in the onset of multimorbidity (16, 17). However, important gaps persist in the understanding of how individual indicators of socioeconomic deprivation and comorbidities affect clinical outcomes after heart transplantation in European universal healthcare systems with free access to multidisciplinary team management programs.
The structure and content of Danish healthcare registers ensure a unique and virtually complete individual-level linkage of data and long-term follow-up (18). Furthermore, the universal healthcare model in Denmark provides health service free of charge to all residents. We used the Scandiatransplant Database (STD) and nationwide health and administrative registers to examine the 20-year prognosis of all heart-transplant recipients in Denmark and the prognostic impact of individual-level SEP and comorbidities.
Matrials and Methods
Setting
The Danish national healthcare system provides tax-financed healthcare for all residents at general practitioners and hospitals as well as reimbursement of prescribed medical therapy. The Civil Registration System (CRS) can unambiguously link up-to-date national health and administrative register data using a unique 10-digit identifier assigned to all residents at birth or upon immigration (18). Denmark has two transplant centers at the University Hospital of Copenhagen and at Aarhus University Hospital.
Data Sources
This study was based on data from: 1) STD, which covers data on all Danish heart-transplant recipients and donors (19), 2) The Danish National Patient Registry (DNPR) (18) containing information on discharge diagnosis according the International Classification of Diseases (ICD-8 and since 1994 ICD-10 codes), along with codes for diagnostic and surgical procedures (18), 3) The Psychiatric Central Research Register (PCRR) containing information on psychiatric diagnoses (18), 4) The Danish National Prescription Registry (NPR) (18), containing data on all redeemed prescriptions at Danish community pharmacies (18). Medical therapies were identified by substance level (Anatomical Therapeutic Chemical [ATC] Classification), 5) The Danish Causes of Death Registry (DCDR) (18), where causes of death are listed as the immediate, underlying, and contributing cause of death (18), 6) CRS including data on vital status, date of birth, gender, and marital status (18), and finally 7) Statistics Denmark (18) covering information from the Education Registry, the Income Statistics Register, and the Integrated Database for Labor Market Research.
This study was approved by the Danish Data Protection Agency (no: 1-16-02-656-18) and the Danish Patient Safety Authority, authorizing access to medical records (no: 3-3013-3173/1).
Study Population and Characteristics
We established a nationwide cohort study including Danish first-time heart-transplant recipients during 1994–2018 recorded in the STD by ICD-10 code (DZ94.1). The index date was defined as the date of the first surgical heart transplantation in the STD. Heart-transplant recipients were followed from index date until 31 December 2018, emigration, or death, whichever occurred first. Recipients undergoing re-transplantation identified in the DNPR (KFQA00, KFQA10) were not censored, since reoperation would be part of the causal pathway of long-term outcome. Information on age, gender, and vital status was retrieved from the CRS (18). Data on donor age and gender mismatch (donor/recipient) were extracted from the SDT.
Age at index date was categorized as 0-20, 21-40, 41-60, and ≥61 years, due to increasing complexities in early, middle, and long-term management post-surgery (20); follow-up time was defined as 0–1, >1–10, and >10 years. The number of recipients alive at end of follow-up was calculated. We collected information on clinically relevant comorbidities by ICD codes registered in the DNPR (18) and PCRR (18) 10 years prior to the index date: Myocardial infarction, angina pectoris, heart failure, heart valve disease, cardiac arrhythmia, congenital heart disease, cardiomyopathy, cardiac inflammation, aortic disease, peripheral arterial disease, cerebrovascular disease, cardiogenic shock and pulmonary edema, diabetes, hypertension, chronic obstructive pulmonary disease, obesity, and psychiatric disorder (Supplementary Table S1). Based on the definition of multimorbidity in other Danish studies (20, 21), we summarized the number of comorbidities 10 years prior to the index date. This Danish algorithm estimates multimorbidity as the co-occurrence of two or more chronic conditions included in the 11 comprehensive chronic disease groups (Supplementary Table S3). Medical treatment was defined as ≥1 redeemed prescription 6 months prior to the index date retrieved from the NPR (18). Polypharmacy was defined as redeeming at least one prescription for ≥5 different cardiovascular agents (18) (Supplementary Table S3).
Individual-Level Socioeconomic Position
Data on individual-level SEP were obtained from Statistics Denmark. Cohabitation status at index date was defined as living alone or cohabiting. We used the highest attained educational level in the calendar year prior to the index date (18) and categorized educational level into five groups: Low (primary and lower secondary education), medium (upper secondary education and academy profession degree), high (bachelor and above), not completed an education (patients age ≤16 years), and missing. We used personal income (pre-tax total) within the calendar year prior to the index date. Based on the annual percentiles in the Danish population, we classified income into percentiles and used the 25th percentile as the cut-off point for low (≤25th percentile) and medium-high (>25th percentile) personal income. Occupational status in the year prior to the index date (18) was grouped into working, non-working (no employment or early retirement), out-of-workforce (state pension, under education), and missing (Supplementary Table S4).
Outcomes
We used the CRS (18) to ascertain date on all-cause mortality during the years following the index date. We also examined cause of mortality using information from the DCDR (18). Cause of mortality was defined by underlying cause and possible cause (immediate cause when available, 1st contributory cause when immediate cause was missing, or 2nd contributory cause when immediate and 1st contributory cause was missing). We generated a list of all documented causes (ICD-10 codes) and divided these into twelve categories: Complications to heart transplantation, multiple organ failure, sudden death, cardiovascular disease, heart failure, cerebrovascular disease, infection, pulmonary disease, malignancy, kidney disease, diabetes, other specified, and not specified (Supplementary Table S5).
The first-time occurrence of hospital admission with a cardiovascular event after the index date was examined (acute myocardial infarction, peripheral arterial disease, cardia arrest, stroke, cardiac inflammation and infection, readmission due to heart failure, graft failure, percutaneous coronary intervention, radiofrequency ablation for atrial fibrillation, cardiac pacemaker, and valve surgery) (Supplementary Table S6). Information was retrieved from the DNPR by primary in-patient diagnosis and surgical procedure codes (18). We investigated the risk of first-time major adverse cardiovascular event (MACE). Composite MACE included readmission due to heart failure, graft failure, percutaneous coronary intervention, acute myocardial infarction, cardiac arrest, and all-cause mortality. To account for potential misclassification of first-time occurrence of hospital admission due to a MACE (especially graft failure due to standard biopsy controls in the first post-transplant year; heart failure, which could follow from prior index date), we conducted a blanking period of 365 days after the index date (Supplementary Table S7).
Statistical Analyses
Baseline data were reported as mean and standard deviation (SD) if normally distributed and as median with 25th‒75th interquartile range (IQR) if skewed continuous data. Categorical data were presented as prevalence (percentage).
Cause of mortality and first-time cardiovascular events were recorded in numbers and percentages. The Kaplan-Meier method was used to compute the risk of all-cause mortality (All). Conditional analyses were performed in recipients who survived the first year (1-year Post-surgery Survival). As supplementary, survival was stratified by time era (1994–2000, 2001–2010, 2011–2018). In addition, the Kaplan-Meier method was used to compute the risk of first-time MACE using the first year after the index date as a blanking period (1-year Post-surgery MACE). Due to Danish law on data protection, first-time acute myocardial infarction (≤5) and cardiac arrest (≤5) were not included in the MACE. However, sensitivity analysis including these events did not change the results. As supplementary, survival and first-time MACE were stratified by gender. To identify the most socially disadvantaged recipients, all socioeconomic factors were dichotomized by the worst quartile or lowest status. Recipients with low educational level (low-degree) were compared to those with medium-higher educational level (medium-high-degree). Recipients <16 years and with missing information on education were not included. Prognostic outcomes were assessed among unemployed (non-working) compared to employed (working, out-of-workforce) recipients. In case of missing information on occupational status, recipients were excluded. Due to the limited sample size, it was not possible to further categorize the exposure variables.
Based on the increasing complexity in long-term management after transplantation (20), we determined the impact of all exposure variables on prognostic outcomes within follow-up intervals: 0–1, >1–10, and >10–20 years. Crude and adjusted hazard ratios (HRs) were computed using Cox Proportional Hazards regression comparing recipients within the dichotomized socioeconomic groups. In multivariable analyses, we adjusted for age, gender, donor age, gender mismatch, hypertension, and diabetes. We evaluated the proportional hazards assumption by visual inspection of log-log plots. Since the median number of comorbidities at baseline was one and less than 2% of the recipients had a psychiatric disorder, these two covariates did not change the results and were thus not included in the regression. We found no indication of any difference between the two Danish transplant centers and transplantation site was not distinguished between in the analyses. A post hoc power analysis was not performed as the utility to inform outcome already observed seems analytically misleading (22). All statistical analyses were performed using SAS statistical software package (version 9.4) and R version 4.1.0 (2021-05-18).
Results
Between 1994 and 2018, first-time heart transplantation was performed in 649 recipients in Denmark (Table 1). Most recipients were male (78%) and 59% were between 41 and 60 years of age at surgery date. Diabetes and hypertension both occurred in 12% of recipients. The median (IQR) number of comorbidities within 10 years prior to transplantation was 1 (1–2). Psychiatric disorder was present in ≤5 of recipients. Median donor age was 41 (IQR, 27–50) and gender mismatch was present in 29%.
TABLE 1
| Total | |
|---|---|
| N = 649 | |
| Gender | |
| Male | 503 (78) |
| Female | 146 (22) |
| Age in years | |
| 0–20 | 67 (10) |
| 21–40 | 117 (18) |
| 41–60 | 381 (59) |
| ≥61 | 84 (13) |
| Donor | |
| Age, median (IQR) | 41 (27–50) |
| Gender mismatch | 118 (29) |
| Follow-up time in years | |
| 0–1 | 97 (15) |
| >1–10 | 296 (46) |
| >10 | 256 (39) |
| Median (IQR) | 7.4 (2.7–13.7) |
| Alive at end of follow-up | 375 (58) |
| Comorbidities (10 years prior to the index date) | |
| Myocardial infarction | 211 (33) |
| Angina Pectoris | 272 (42) |
| Heart failure | 547 (84) |
| Heart valve disease | 71 (11) |
| Cardiac arrhythmia | 307 (47) |
| Congenital heart disease | 70 (11) |
| Cardiomyopathy | 434 (67) |
| Cardiac inflammation | 66 (10) |
| Aortic disease | —a |
| Peripheral arterial disease | 10 (2) |
| Cerebrovascular disease | 61 (9) |
| Cardiogenic shock and pulmonary edema | 57 (9) |
| Diabetes | 77 (12) |
| Hypertension | 80 (12) |
| Chronic obstructive pulmonary disease | 69 (11) |
| Obesity | 21 (3) |
| Mental disease | —a |
| Multimorbidity (10 years prior to the index date) | |
| Number of chronic diseases, median (IQR) | 1 (1–2) |
| Cardiovascular polypharmacy (6 months prior to the index date) b | |
| Prescribed medications ≥5 | 348 (54) |
| Cohabitation status | |
| Living alone | 281 (43) |
| Cohabitation | 368 (57) |
| Highest obtained educational degree | |
| Low (primary and lower secondary education) | 193 (30) |
| Medium (upper secondary education and academy profession) | 283 (44) |
| High (bachelor and above) | 116 (18) |
| Not completed education (patients age ≤16 years) | 40 (6) |
| Missing | 17 (3) |
| Personal income group | |
| Low income (≤25th percentile) | 134 (21) |
| Medium-high income (>25th percentile) | 515 (79) |
| Occupational status | |
| Working | 300 (46) |
| Non-working | 27 (4) |
| Out-of- workforce (state pension, under education) | 300 (46) |
| Missing | 22 (3) |
Baseline characteristic in heart-transplant recipients.
Values are n (%).
Due to data protection (<5 patients).
Data available since 1995 in the Danish National Prescription Registry.
Outcomes
Twenty-year survival curves for all-cause mortality are displayed in Figure 1. The half-time survival was 15.6 years (95% confidence interval [CI] 13.8–17.5) and 17.6 years when conditional on 1-year survival (95% CI 16.2–19.1) (Supplementary Figures S1–S4). The leading underlying causes of mortality were heart failure (25%), cardiovascular disease (18%), and malignancy (18%) (Table 2). The three cardiovascular first-time events with the highest incidence (within 1–20 years post-surgery) were graft failure (42%), readmission due to heart failure (14%), and percutaneous coronary intervention (21%) (Table 3). Both acute myocardial infarction and cardiac arrest occurred in ≤5 of recipients. Approximately half of the heart transplant recipients were at risk of a first-time MACE within an 11-year period after the index date among those surviving to at least 1-year (Figure 2).
FIGURE 1
TABLE 2
| Overall (N = 649) | Underlying cause | Possible causea |
|---|---|---|
| n = 274 | n = 274 | |
| Complications of heart transplantation | —b | 16 (6) |
| Multiple organ failure | —b | 19 (7) |
| Sudden deaths | —b | 25 (9) |
| Cardiovascular disease | 48 (18) | 18 (7) |
| Heart failure | 68 (25) | 32 (12) |
| Cerebrovascular disease | —b | 15 (5) |
| Infection | 12 (4) | 18 (7) |
| Pulmonary disease | —b | 23 (8) |
| Malignancy | 48 (18) | 22 (8) |
| Kidney disease | —b | 12 (4) |
| Diabetes | —b | —b |
| Other specified | —b | —b |
| Not specified | 55 (20) | 39 (14) |
Cause of mortality in heart-transplant recipients.
Recipients were followed after heart transplantation (index day) and until 31 December 2018, emigration, or mortality, whichever occurred first.
Values are n (%).
Immediate cause when it is available; 1st contributory cause when immediate cause is missing; 2nd contributory cause when immediate and 1st contributory cause is missing.
Due to data protection (<5).
TABLE 3
| Overall (N = 649) | n = 330 |
|---|---|
| Acute myocardial infarction | —a |
| Peripheral arterial disease | 11 (3) |
| Cardiac arrest | —a |
| Stroke | 10 (3) |
| Cardiac inflammation and infection | —a |
| Readmission due to heart failure | 47 (14) |
| Graft failure | 140 (42) |
| Percutaneous Coronary Intervention | 68 (21) |
| Radiofrequency ablation for atrial fibrillation | —a |
| Cardiac pacemaker | 18 (6) |
| Valve surgery | 11 (3) |
First-time cardiovascular event in heart-transplant recipients.
Recipients were followed from day +365 after heart transplantation (index day) and until 31 December 2018, emigration, or death, whichever occurred first.
Values are n (%).
Due to data protection (<5 events).
FIGURE 2
Individual-Level Socioeconomic Position
Adjusted HRs for all-cause mortality by socioeconomic factors and in different follow-up intervals are presented in Figure 3. Low educational level was associated with all-cause mortality within the period 10–20 years after heart transplantation (HR 1.95, 95% CI 1.19–3.19); otherwise we found no associations between socioeconomic factors and all-cause mortality (Supplementary Table S8). In contrast, we observed SEP-related associations with first-time MACE (Figure 4). During both >1–10 years and >10–20 years after the index date, low educational level was associated with first-time MACE. Low income was associated with first-time MACE within >1–10 years after the index date (HR 1.81, 95% CI 1.02–3.22). Cohabitation status was not significantly associated with first-time MACE during follow-up intervals. However, although it did not reach significance there was a suggestion that living alone was associated with a higher risk of first-time MACE within >1–10 years (HR 1.46, 95% CI 0.98–2.17). No associations between occupational status and first-time MACE were documented (Figure 4) (Supplementary Table S9).
FIGURE 3
FIGURE 4
Discussion
In this nationwide cohort study comprising all Danish first-time heart-transplant recipients during a 20-year period, the half-time survival estimate was 15.6 years. The highest prevalence of first-time cardiovascular events was graft failure. This study revealed two major findings: 1) low educational level at index date was associated with higher risk of all-cause mortality within 10–20 years after heart transplantation and 2) low educational level, low income, and a suggestion towards living alone were associated with higher risk of first-time MACE within 1–10 years post-transplant.
In a Scandinavian cohort (1983–2009) of heart-transplant recipients (n = 2293; 8% <18 years), the half-time survival was estimated to 13.2 years (19) and 15.3 years when conditional survival was set at 1-year. We demonstrated an excellent half-time survival (15.6 years) as well as 1-year conditional survival (17.6) when compared with internationally published data. This may be attributed to heart-transplant survival consistently improving over the last 30 years and has been described as the era effect. Heart transplantation in Denmark was initiated later than in the United States and other European countries (, 19, 23). This is supported by our supplementary survival curves in Danish heart-transplant recipients stratified by time period (1994–2000, 2001–2010, 2011–2018). In the current study, we found that the three cardiovascular first-time events with the highest incidence were graft failure, readmission due to heart failure, and percutaneous coronary intervention. Approximately half of the heart-transplant recipients were at risk of a first-time MACE within 11 years after transplantation conditional on survival of at least 1-year. Our findings consolidate that graft failure and rejection remain the leading causes of mortality post-transplant (, ). Cardiac allograft vasculopathy is the main reason for allograft failure, and percutaneous coronary intervention is usually considered a palliative treatment because of the progressive nature of vasculopathy (24). In addition, a recent study based on the nationwide readmission database in the United States reported that heart failure is one of the main primary unplanned diagnoses causing readmission after heart transplantation (25). We were not able to establish whether gender influence survival and first-time MACE curves since only 23% of the recipients were female (Supplementary Figures S3, S4). Scandinavian results on long-term follow-up after heart transplantation (n = 2293) have documented no significant difference in survival when stratified by gender (p = 0.44) (19). However, this issue warrants further investigation.
Several previous studies have linked SEP to prognostic outcomes in heart-transplant recipients. A nationwide follow-up study in England, including 2,384 adult heart transplant-recipients (1995–2014) demonstrated that the most socioeconomically disadvantaged recipients had a 27% higher risk-adjusted 19-year overall mortality (HR 1.27, 95% CI 1.04–1.55). The United Kingdom multiple deprivation index was used to measure SEP (). Similarly, a study () using the UNOS database in 36,736 adult (≥18 years) first-time heart-transplant recipients (1994–2014) found that college educated patients had an 18% reduced rate of deaths. Moreover, lowest SEP (index of seven SEP indicators) confers higher unadjusted risk of post-transplant hospitalization (HR 1.13), rejection (HR 1.28), infection (HR 1.10), and ischemic event (HR 1.26) (). Another UNOS-based study including 5,125 primarily pediatric heart transplant recipients (2000–2011) reflected that risk adjusted survival was poorer in groups with a low SEP (HR 1.41, 95% CI 1.10–1.80) (26). Findings from a single-center Boston study among first-time heart transplant recipients (n = 520) conducted between 1996 and 2005 supported that low SEP (score of six variables) was associated with higher adjusted risk of graft loss (HR 1.5, 95% CI 1.0–2.4) (). Findings from a previous UNOS analysis in left ventricular assist devices (LVAD) recipient’s ≥18 years (n = 3361) waiting for heart transplantation demonstrated that recipients with lower SEP (index of seven SEP indicators) had an early and sustained decreased adjusted post-transplant survival (lowest quartile: HR 0.57, 95% CI 0.39–0.82; highest quartile: HR 0.68, 95% CI 0.48–0.95 (). Moreover, an analysis of the UNOS database including 33,893 adult heart-transplant recipients suggested an increased risk of mortality or re-transplantation (Adjusted p <0.001) associated with public health insurance status (Medicaid or Medicare versus private) (). Research also based on the UNOS database studying a population (n = 20,676) of heart transplant recipients >17 years showed that Medicare and Medicaid insurances were associated with lower 10-year mortality risk (18%, 33%, respectively) than private insurance (). In addition, multivariable analyses found that college-education decreased risk of mortality with 11% (). In contrast to most previous studies using area-based social deprivation indexes or under-insurance status, we examined socioeconomic factors by individual and complete register-based single indicators of social vulnerability. Between 1 and 10 years post-surgery in particular, we observed a modest SEP gradient in the risk of a first-time MACE in heart transplant recipients. Remarkably, our results reflect that low educational attainment could be the most influential factor on both mortality and MACE, whereas personal income only influenced MACE. A recent single-center Danish study including 325 first-time heart transplant recipients (79% male and 69% between 41 and 60 years) described a lower median number of redeemed medical prescriptions during 15 years of follow-up in heart-transplant recipients within the lowest income group or if living alone (20). The association between income and prognosis could thus also be partly driven by an economic gradient in use of the prescribed medical treatment after heart transplantation. In line with the current understanding (, , ), it seems possible that even in a country with free access to multidisciplinary team management programs, educationally and economically disadvantaged heart transplant recipients could have an increased risk of non-adherence to immunosuppressive treatment, inadequate self-management skills, experience health disparities, and missed healthcare delivery; thus, graft failure and all-cause mortality are more likely in these recipients. However, our results indicate that the individual-level SEP impacts the middle follow-up period 1–10 years after transplantation. The most likely explanation for this is that socioeconomic disparities narrow over time after heart transplantation due to the role of the multidisciplinary team management identifying barriers to medical adherence and engaging patients to follow health recommendations. In accordance with the single-center Danish study, we believe that living alone may negatively influence on pharmalogical self-care. The lack of association between living alone and prognosis may be a result of the small sample size.
Our study also included information on comorbidities and chronic mental diseases 10 years prior to heart transplantation. Since the median number of comorbidities at baseline was one and less than 1% of the recipients had a psychiatric disorder, multimorbidity and psychiatric disorder were too rare to allow for further analyses of interactions. This may be explained by careful recipient selection based on pre- and post-transplant life expectancy, which reflects the recipient’s pre-operative psychosocial status and comorbidity burden (, ).
Remarkably, a prior UNOS study (27) in the United States (2001–2014) investigated the effect of non-working of heart transplant recipients (n = 23.228, >18 years) on survival. An adjusted analysis demonstrated a 5% and 10% decrease in 5- and 10-year mortality, respectively. Our study did not reveal any influence of occupational status. The most likely explanation is that our cohort included recipients >65 years (age at receiving state pension in Denmark) as well as the early retirement status of chronic end-stage heart failure recipients.
Although the Danish healthcare system appears to ensure easy access to multidisciplinary team management programs and fully funded immunosuppressive and medical treatment, our results support that in mainly educationally and economically disadvantaged recipients, the long-term prognosis of heart-transplant recipients is affected. This study contributes with knowledge to target long-term healthcare strategies for socially disadvantaged heart-transplant recipients across the world (16, 17). Our data suggests the need to focus on socioeconomic factors and their influence on both adherence and rehabilitation to support adequate self-management, self-efficacy, and health literacy after heart transplantation (). The development of new mobile health devises (mHealth) in the field of transplantation has immense potential to facilitate healthcare service and implement more individualized education and management programs (28, 29). Further studies are needed to design and address delivery of more socially differentiated multidisciplinary team management programs for this patient group.
The setting in our study, including all heart transplant recipients in Denmark with long-term follow-up and individual accurate data linkage within a uniform healthcare system, reduced selection and recall bias. A critical limitation was that data in the DCDR (18) were not validated. Thus, the diagnosis of both underlying and contributory causes depends on the decision of the individual physician. We used a simple disease count algorithm to estimate the degree of multimorbidity. Thus, the relative severity of disease combinations was not assessed, and residual confounding could thus occur. Another limitation is the lack of precise temporality between baseline SEP and all-cause mortality or MACE, which does not allow inference from the identified observations. Notably, the combined MACE has not been validated. However, the component outcomes were validated in the general populations (18). Even though we adjusted our analysis for important confounding factors, residual confounding cannot be ruled out, since important clinical risk factors, blood sample measurements, and surgical procedure data were not available. Due to the small sample size, our reported associations should be supported in future large-scale observational studies.
We found that in first-time heart transplant recipients, the half-time survival was 15.6 years during a 20-year period. Low levels of education and income were associated with a poorer prognosis after surgery despite selection during the assessment process leading to heart transplantation.
Statements
Data availability statement
Study data, statistical plan, and log-files can be made available through proposal to the Project Database (ID: 707738) at Statistics Denmark. https://www.dst.dk/en/TilSalg/Forskningsservice.
Ethics statement
Ethical review and approval was not required for the study on human participants in accordance with the local legislation and institutional requirements. Written informed consent from the participants’ legal guardian/next of kin was not required to participate in this study in accordance with the national legislation and the institutional requirements.
Author contributions
RM, HE, and BBL designed the study. RM collected the data. RM, HE, and BL directed data management and analysis, which were carried out by IB and EH-P. All authors participated in the discussion and interpretation of results. RM, HE, and BL organized the writing and RM wrote the initial draft. All authors critically revised the manuscript and approved the final version.
Funding
This study was supported by grants from The Karen Elise Jensen Foundation (July 2019) and The Helse Foundation (20-B-0155). The sponsor had no role in study design, data collection, analysis or interpretation of the data, writing of the manuscript, or in the decision to submit the manuscript for publication.
Acknowledgments
We thank the Scandiatransplant Database for providing data for this study.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontierspartnerships.org/articles/10.3389/ti.2023.10976/full#supplementary-material
Abbreviations
ATC, anatomical therapeutic chemical; CRS, civil registration system; DCDR, Danish causes of death registry; DNPR, Danish national patient registry; MACE, major adverse cardiovascular event; NPR, Danish national prescription registry; PCRR, psychiatric central research registry; SEP, socioeconomic position; STD, Scandiatransplant database.
References
1.
MehraMRCanterCEHannanMMSemigranMJUberPABaranDAet alThe 2016 International Society for Heart Lung Transplantation Listing Criteria for Heart Transplantation: A 10-year Update. J Heart Lung Transplant (2016) 35(1):1–23. 10.1016/j.healun.2015.10.023
2.
McDonaghTAMetraMAdamoMGardnerRSBaumbachABöhmMet al2021 ESC Guidelines for the Diagnosis and Treatment of Acute and Chronic Heart Failure. Eur Heart J (2021) 42(36):3599–726. 10.1093/eurheartj/ehab368
3.
KhushKKCherikhWSChambersDCHarhayMOHayesDJr.HsichEet alThe International Thoracic Organ Transplant Registry of the International Society for Heart and Lung Transplantation: Thirty-Sixth Adult Heart Transplantation Report - 2019; Focus Theme: Donor and Recipient Size Match. J Heart Lung Transplant (2019) 38(10):1056–66. 10.1016/j.healun.2019.08.004
4.
LundLHEdwardsLBDipchandAIGoldfarbSKucheryavayaAYLevveyBJet alThe Registry of the International Society for Heart and Lung Transplantation: Thirty-Third Adult Heart Transplantation Report-2016; Focus Theme: Primary Diagnostic Indications for Transplant. J Heart Lung Transplant (2016) 35(10):1158–69. 10.1016/j.healun.2016.08.017
5.
CostanzoMRDipchandAStarlingRAndersonAChanMDesaiSet alThe International Society of Heart and Lung Transplantation Guidelines for the Care of Heart Transplant Recipients. J Heart Lung Transplant (2010) 29(8):914–56. 10.1016/j.healun.2010.05.034
6.
WaydaBClemonsAGivensRCTakedaKTakayamaHLatifFet alSocioeconomic Disparities in Adherence and Outcomes after Heart Transplant: A UNOS (United Network for Organ Sharing) Registry Analysis. Circ Heart Fail (2018) 11(3):e004173. 10.1161/CIRCHEARTFAILURE.117.004173
7.
EvansJDKaptogeSCaleyachettyRDi AngelantonioELewisCParameshwarKJet alSocioeconomic Deprivation and Survival after Heart Transplantation in England: An Analysis of the United Kingdom Transplant Registry. Circ Cardiovasc Qual Outcomes (2016) 9(6):695–703. 10.1161/CIRCOUTCOMES.116.002652
8.
ClerkinKJGaranARWaydaBGivensRCYuzefpolskayaMNakagawaSet alImpact of Socioeconomic Status on Patients Supported with a Left Ventricular Assist Device: An Analysis of the UNOS Database (United Network for Organ Sharing). Circ Heart Fail (2016) 9(10):e003215. 10.1161/CIRCHEARTFAILURE.116.003215
9.
SinghTPGivertzMMSemigranMDenofrioDCostantinoFGauvreauK. Socioeconomic Position, Ethnicity, and Outcomes in Heart Transplant Recipients. Am J Cardiol (2010) 105(7):1024–9. 10.1016/j.amjcard.2009.11.015
10.
AllenJGWeissESArnaoutakisGJRussellSDBaumgartnerWAShahASet alInsurance and Education Predict Long-Term Survival after Orthotopic Heart Transplantation in the United States. J Heart Lung Transplant (2012) 31(1):52–60. 10.1016/j.healun.2011.07.019
11.
DewMARosenbergerEMMyaskovskyLDiMartiniAFDeVito DabbsAJPoslusznyDMet alDepression and Anxiety as Risk Factors for Morbidity and Mortality after Organ Transplantation: A Systematic Review and Meta-Analysis. Transplantation (2015) 100(5):988–1003. 10.1097/TP.0000000000000901
12.
DewMADiMartiniAFDobbelsFGradyKLJowsey-GregoireSGKaanAet alThe Approach to the Psychosocial Evaluation of Cardiac Transplant and Mechanical Circulatory Support Candidates. Curr Heart Fail Rep (2019) 16(6):201–11. 10.1007/s11897-019-00443-0
13.
BurkerBSGullestadLGudeEHavikOERelbo AuthenAGrovIet alThe Predictive Value of Depression in the Years after Heart Transplantation for Mortality during Long-Term Follow-Up. Psychosomatic Med (2019) 81(6):513–20. 10.1097/PSY.0000000000000702
14.
SchiøtzMLStockmarrAHøstDGlümerCFrølichA. Social Disparities in the Prevalence of Multimorbidity - A Register-Based Population Study. BMC Public Health (2017) 17(1):422. 10.1186/s12889-017-4314-8
15.
LawrenceDKiselySPaisJ. The Epidemiology of Excess Mortality in People with Mental Illness. Can J Psychiatry Revue canadienne de psychiatrie (2010) 55(12):752–60. 10.1177/070674371005501202
16.
BarnettKMercerSWNorburyMWattGWykeSGuthrieB. Epidemiology of Multimorbidity and Implications for Health Care, Research, and Medical Education: a Cross-Sectional Study. Lancet (London, England) (2012) 380(9836):37–43. 10.1016/S0140-6736(12)60240-2
17.
PathiranaTIJacksonCA. Socioeconomic Status and Multimorbidity: a Systematic Review and Meta-Analysis. Aust New Zealand J Public Health (2018) 42(2):186–94. 10.1111/1753-6405.12762
18.
SchmidtMSchmidtSAJAdelborgKSundbollJLaugesenKEhrensteinVet alThe Danish Health Care System and Epidemiological Research: from Health Care Contacts to Database Records. Clin Epidemiol (2019) 11:563–91. 10.2147/CLEP.S179083
19.
DellgrenGGeiranOLemströmKGustafssonFEiskjaerHKoulBet alThree Decades of Heart Transplantation in Scandinavia: Long-Term Follow-Up. Eur J Heart Fail (2013) 15(3):308–15. 10.1093/eurjhf/hfs160
20.
MolsREBakosIChristensenBHorváth-PuhóELøgstrupBBEiskjærH. Influence of Multimorbidity and Socioeconomic Factors on Long-Term Cross-Sectional Health Care Service Utilization in Heart Transplant Recipients: A Danish Cohort Study. J Heart Lung Transplant (2022) 41:527–37. 10.1016/j.healun.2022.01.006
21.
JensenLFPedersenAFAndersenBVestergaardMVedstedP. Non-participation in Breast Cancer Screening for Women with Chronic Diseases and Multimorbidity: a Population-Based Cohort Study. BMC cancer (2015) 15:798. 10.1186/s12885-015-1829-1
22.
ZhangYHedoRRiveraARullRRichardsonSTuXM. Post Hoc power Analysis: Is it an Informative and Meaningful Analysis?Gen Psychiatry (2019) 32(4):e100069. 10.1136/gpsych-2019-100069
23.
HessNRSeeseLMMathierMAKeeblerMEHickeyGWMcNamaraDMet alTwenty-year Survival Following Orthotopic Heart Transplantation in the United States. J Card Surg (2021) 36(2):643–50. 10.1111/jocs.15234
24.
ShahHPatelSSolankiSWudexiISinghJMullangiSet alClinical Characteristics and in Hospital Outcomes of Heart Transplant Recipients Undergoing Percutaneous Coronary Intervention: Insights from the National Inpatient Sample. Catheterization Cardiovasc interventions (2020) 96(6):E585–E592. 10.1002/ccd.29184
25.
AlvarezPABriasoulisAMalikAH. Trends, Risk Factors, and Mortality of Unplanned 30-day Readmission after Heart Transplantation. Am J Cardiol (2021) 154:130–3. 10.1016/j.amjcard.2021.06.002
26.
DaviesRRRussoMJReinhartzOMaedaKRosenthalDNChinCet alLower Socioeconomic Status Is Associated with Worse Outcomes after Both Listing and Transplanting Children with Heart Failure. Pediatr Transplant (2013) 17(6):573–81. 10.1111/petr.12117
27.
RaviYLellaSKCopelandLAZolfaghariKGradyKEmaniSet alDoes Recipient Work Status Pre-transplant Affect post-heart Transplant Survival? A United Network for Organ Sharing Database Review. J Heart Lung Transplant (2018) 37(5):604–10. 10.1016/j.healun.2018.01.1307
28.
FlemingJNPollockMDTaberDJMcGillicuddyJWDiamantidisCJDochertySLet alReview and Evaluation of mHealth Apps in Solid Organ Transplantation: Past, Present, and Future. Transplant direct (2022) 8(3):e1298. 10.1097/TXD.0000000000001298
29.
Gomis-PastorMMirabet PerezSRoig MinguellEBrossa LoidiVLopez LopezLRos AbarcaSet alMobile Health to Improve Adherence and Patient Experience in Heart Transplantation Recipients: The mHeart Trial. Healthcare (Basel, Switzerland) (2021) 9(4):463. 10.3390/healthcare9040463
Summary
Keywords
mortality, heart transplantation, prognosis, individual-level, socioeconomic position
Citation
Mols RE, Løgstrup BB, Bakos I, Horváth-Puhó E, Christensen B, Witt CT, Schmidt M, Gustafsson F and Eiskjær H (2023) Individual-Level Socioeconomic Position and Long-Term Prognosis in Danish Heart-Transplant Recipients. Transpl Int 36:10976. doi: 10.3389/ti.2023.10976
Received
16 October 2022
Accepted
10 March 2023
Published
22 March 2023
Volume
36 - 2023
Updates
Copyright
© 2023 Mols, Løgstrup, Bakos, Horváth-Puhó, Christensen, Witt, Schmidt, Gustafsson and Eiskjær.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Rikke E. Mols, rikkmols@rm.dk
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.